Evaluation of measurement uncertainty from in-house precision and recovery data
Contents
 Following the publication of ISO/IEC 17025 in 2017, the community has placed greater emphasis on the reliable evaluation of measurement uncertainty. Nowadays, laboratories are expected to be able to use measurement uncertainty in managing and reporting the risk of false decisions regarding the conformity of tested items.
Following the publication of ISO/IEC 17025 in 2017, the community has placed greater emphasis on the reliable evaluation of measurement uncertainty. Nowadays, laboratories are expected to be able to use measurement uncertainty in managing and reporting the risk of false decisions regarding the conformity of tested items.
Given the guidance available on the evaluation of measurement uncertainty produced by Eurachem, Eurolab, and Nordtest, it was considered necessary to develop additional material to help laboratories accurately determine measurement uncertainties.
This Eurachem/CITAC guide is intended to be read in conjunction with the Eurachem guide "Quantifying uncertainty in Analytical Measurement" (QUAM). It discusses the evaluation of measurement uncertainty from in-house validation data, adding detail to the general guidance in QUAM. Key topics include modelling how measurement uncertainty varies with concentrations, and how to handle divergent recovery values from different reference materials. Laboratories now have access to simple algorithms for assessing measurement uncertainty, but for situations where these are insufficient to meet performance requirements, more advanced solutions are provided.
The guide is organised as a tutorial and features numerous practical examples to support its application
Availability
This guidance is available in the following languages:
- Download the guide in English (published 2026-03-15) (pdf, 2.4 Mb).
Citation
This publication should be cited* as:
R Bettencourt da Silva (ed.) Eurachem/CITAC Guide: Evaluation of measurement uncertainty from in-house precision and recovery data. First Edition, Eurachem (2026). ISBN 978-972-9348-30-3. DOI: https://doi.org/10.56526/2026.0005. Available from http://www.eurachem.org
*Subject to journal requirements
Translations
Translation into other languages is permitted for members of Eurachem. Other offers of translation should be directed to the Eurachem Secretariat for permission. The Eurachem policy on maintenance and development of Eurachem guidance, available on the Policies page, gives further information on translation.



 The aim of this guide is to provide laboratories with guidance on best practice for the analytical operations they carry out. The guidance covers both qualitative and quantitative analysis carried out on a routine or non-routine basis.
The aim of this guide is to provide laboratories with guidance on best practice for the analytical operations they carry out. The guidance covers both qualitative and quantitative analysis carried out on a routine or non-routine basis. Guidance is given on how to validate a measuring procedure that includes the primary sampling (VaMPIS). It is a supplement to existing Eurachem guidance on “The Fitness for Purpose of Analytical Methods” and “Measurement Uncertainty arising from Sampling”. The overall aim is to extend the concept of ‘validation of a measurement procedure’ beyond the validation of the analytical method (or procedure) alone, in order to include the primary sampling (and physical sample preparation) within the validation process.
Guidance is given on how to validate a measuring procedure that includes the primary sampling (VaMPIS). It is a supplement to existing Eurachem guidance on “The Fitness for Purpose of Analytical Methods” and “Measurement Uncertainty arising from Sampling”. The overall aim is to extend the concept of ‘validation of a measurement procedure’ beyond the validation of the analytical method (or procedure) alone, in order to include the primary sampling (and physical sample preparation) within the validation process.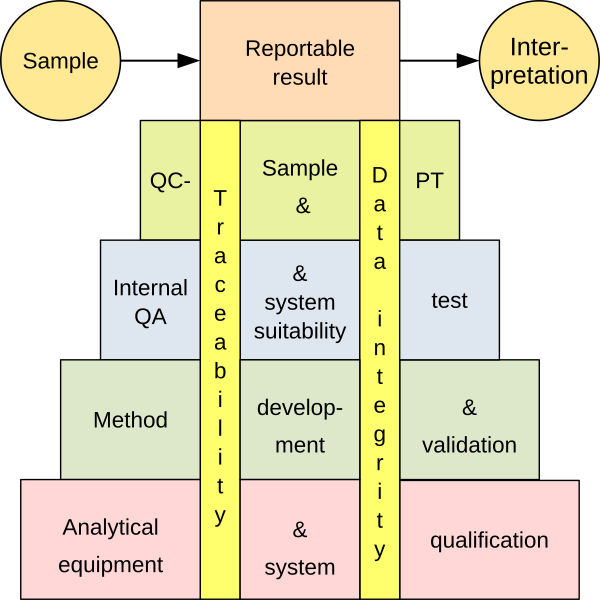 A wide variety of analytical equipment is used in analytical laboratories, ranging from simple apparatus to complex computer-based systems, to collect data that helps to obtain a reportable result. Many of these pieces of equipment combine a measurement function with software control. There are many ways to demonstrate that an equipment is qualified and under control, including qualification, calibration, validation and maintenance. To ensure ‘fitness for purpose’, an integrated approach based on risk assessment is recommended.
A wide variety of analytical equipment is used in analytical laboratories, ranging from simple apparatus to complex computer-based systems, to collect data that helps to obtain a reportable result. Many of these pieces of equipment combine a measurement function with software control. There are many ways to demonstrate that an equipment is qualified and under control, including qualification, calibration, validation and maintenance. To ensure ‘fitness for purpose’, an integrated approach based on risk assessment is recommended.